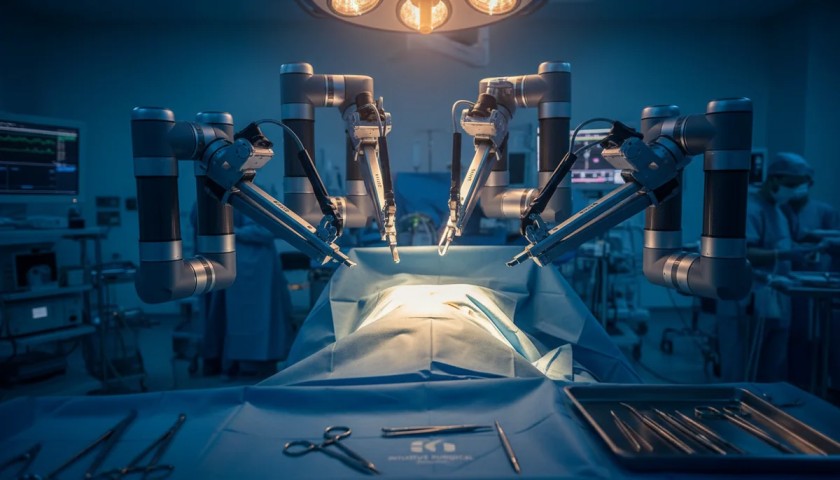
Johnson & Johnson announced results from the first clinical study of the investigational OTTAVA™ Robotic Surgical System – a prospective, multicenter clinical study evaluating the safety and performance of the system in Roux-en-Y gastric bypass procedures. The results were presented at the 2026 American Society for Metabolic and Bariatric Surgery (ASMBS) Annual Meeting (Abstract #4153).
In the 30-patient cohort, the study met its primary safety and performance endpoints through 30 days post-procedure, and investigators completed all procedures robotically on OTTAVA™ without conversion to a non-robotic approach. Average weight loss by 30 days after surgery was 30 pounds. These clinical data, together with preclinical testing, were used to support an application to the U.S. Food and Drug Administration (FDA) for De Novo classification targeting an indication covering multiple procedures in general surgery within the upper abdomen, such as gastric bypass, gastric sleeve, small bowel resection and hiatal hernia repair.
“The data show encouraging evidence regarding the safety and performance of the OTTAVA™ Robotic Surgical System in Roux-en-Y gastric bypass procedures,” said Erik Wilson*, M.D., Chief of Minimally Invasive and Elective General Surgery, UTHealth Houston, Director of Bariatric Surgery, Memorial Hermann-Texas Medical Center, and lead investigator for the clinical study. “Continued innovation in bariatric surgery is important to patient care, and I am pleased to present these initial clinical results.”
Addressing Bariatric Complexities
In the U.S. alone, more than two in five adults have obesity.1 Metabolic and bariatric surgical procedures are effective, long-term treatment options for weight loss management. Many of these procedures, including Roux-en-Y gastric bypass, are increasingly performed robotically.2 Roux‑en‑Y gastric bypass is an essential intervention in the management of obesity, with recent research indicating the treatment is most effective and durable for severe obesity.3,4 The operation is technically demanding relative to other bariatric procedures – requiring both restrictive and reconstructive steps that typically span multiple abdominal quadrants and leveraging a broad spectrum of surgical tasks.
"This is an exciting milestone for our program as we share the role of OTTAVA™ as a robotic surgical system with a novel architecture supporting patient treatment during the clinical trial,” said Hani Abouhalka, Company Group Chair, Surgery, MedTech, Johnson & Johnson. “This work also underscores our long-term commitment to bariatric surgeons and to equipping clinicians with a broad portfolio of surgical technologies to address the complexity of obesity care. Across open, laparoscopic, and robotic surgery, we remain focused on translating innovation into real-world solutions that help surgeons broaden access to precision surgical care and improve outcomes for patients."
Designed for Optimized OR Workflows
OTTAVA™ is designed as a multi-specialty soft-tissue surgery robot, built to enable a broad range of procedures across multiple specialties, to be supported with multiple clinical trials. The system’s novel architecture incorporates four robotic arms integrated into a standard size surgical table, removing the need for a separate boom or carts.
In this study, OTTAVA™ was installed and used successfully across operating rooms (ORs) at all six participating hospitals. OR sizes ranged from approximately 243 to 694 sq. ft. In five of the six sites, procedures were performed in ORs that previously had not been used for robotic surgery, including rooms historically considered challenging for robotic systems due to space constraints.
“Our progress with OTTAVA™ relies on rigorous preclinical work, disciplined data collection in the clinic, and tight collaboration with the surgeons who use it – especially the investigators in the FORTE clinical study. These results further illustrate how novel architecture supports a differentiated approach where it matters most: in real operating rooms, with real patients,” said Peter Schulam, M.D., Ph.D., Chief Scientific Officer, MedTech, Johnson & Johnson. “At each step – from bench to preclinical and now human trial – we have observed that this system can allow more hospital ORs to accommodate robotics. These results demonstrate that the novel architecture can integrate into existing infrastructure and remove practical barriers to broader adoption."